MoCRA recordkeeping for Shopify cosmetic brands — without spreadsheets.
Centralize safety substantiation per SKU. Never miss the 15 business-day serious adverse event clock. Export consistent documentation packs on demand.
- Built around the 15 business-day serious adverse event clock
- Exports include an audit manifest (versions + timestamps)
- No filings, no legal advice — just clean recordkeeping
Built for
US-selling Shopify cosmetics, skincare, and haircare brands
Not for
Enterprise QMS, filing services, or public complaint intake
Get Early Access
Be the first to know when we launch.
Clean interfaces. Organized records. Clear exports.
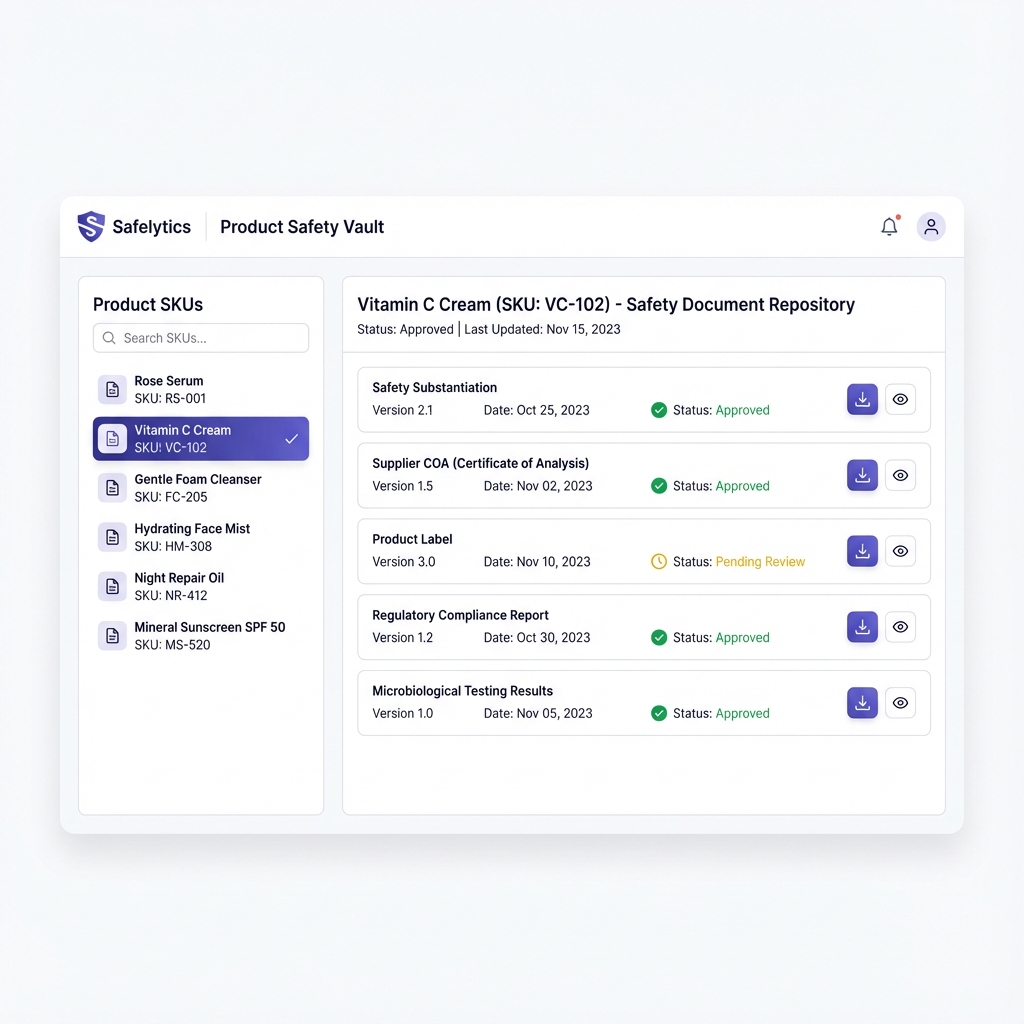
Product Safety Vault
SKU-linked documents with version history
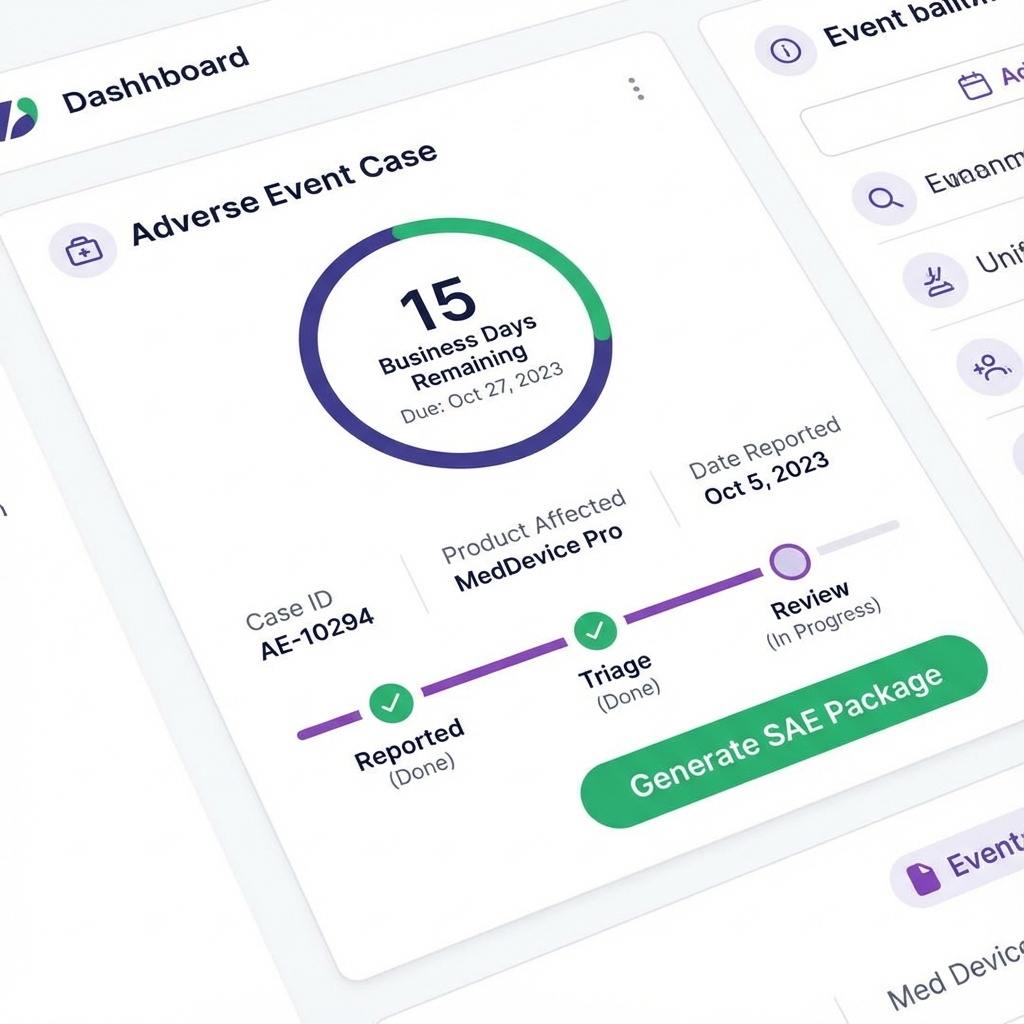
Adverse Event Case
15 business-day clock with due-by date tracking
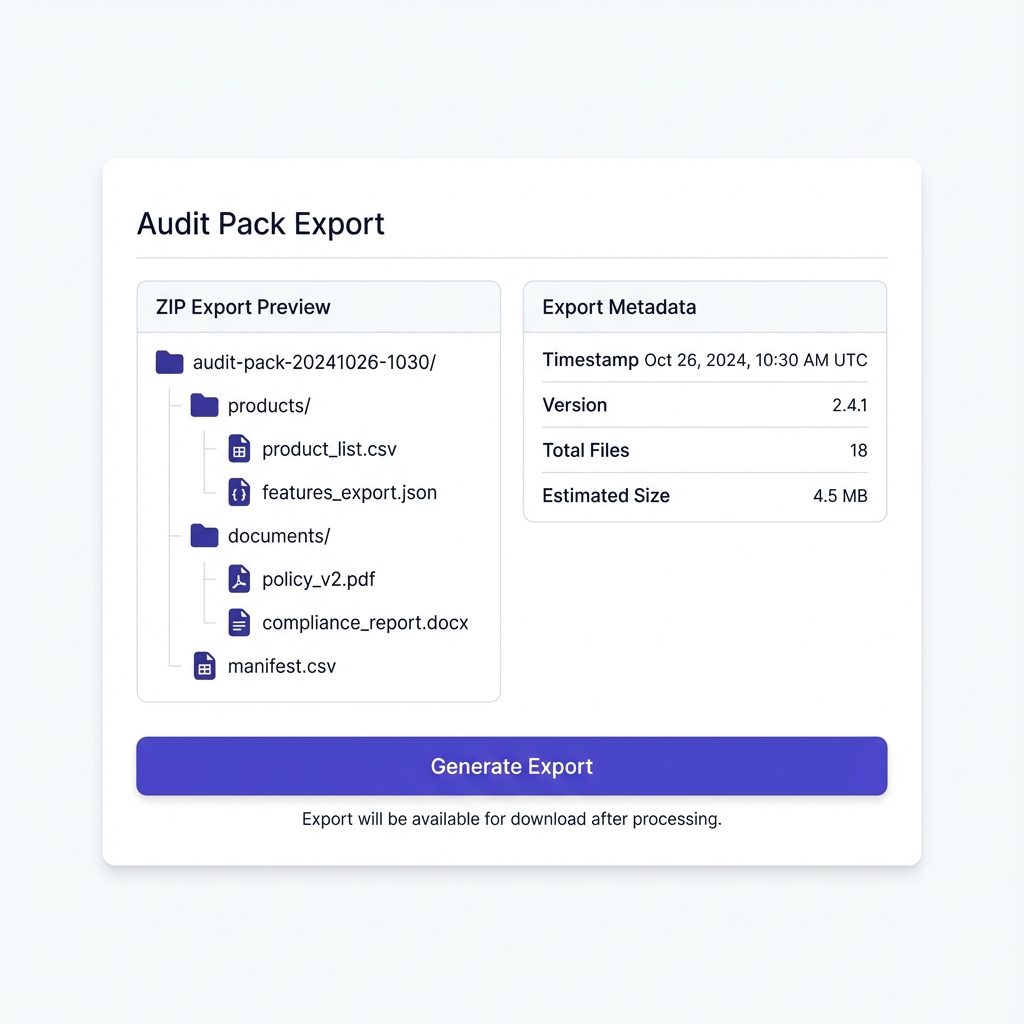
Audit Pack Exports
Manifest + deterministic ZIP structure
Everything you need for MoCRA readiness
Purpose-built tools to help Shopify cosmetic brands organize the documentation they need for FDA's Modernization of Cosmetics Regulation Act.
Product Safety Vault
Organize labels, safety substantiation docs, and supplier documentation per SKU. Structured, versioned records — exportable with an audit manifest (versions + timestamps).
Adverse Event Log
Internal-only workflow for product-related issues. Track the 15 business-day serious adverse event reporting clock. Clear due-by dates, no calendar-day ambiguity.
One-click Audit Pack Export
Generate deterministic ZIP packages on demand. Every export includes a manifest (CSV/HTML index) with versions and timestamps (checksums planned) for full traceability.
MoCRA Profile Tracker
Track facility registration status (renewed every 2 years) and product listing updates (annual, including discontinuations) — plus upload proof docs and get deadline reminders. We do not file anything on your behalf.
SAE Submission Package Export
Generate a Form 3500A-ready packet with label + attachments for serious adverse event reporting — including a checklist for follow-up info received within 1 year (also due within 15 business days). You submit through official FDA channels — we just prepare the package.
Privacy-First
No tracking cookies. We hash IPs for abuse prevention only. Your data stays yours.
Shopify-Native
Built from the ground up for Shopify cosmetic brands. No generic SaaS complexity.
Deterministic Exports
Consistent exports every time. Audit manifest includes versions, timestamps, and file checksums.
Frequently Asked Questions
Important information about what MoCRA Ops Kit does — and doesn't do.
Registration & Listing: You submit via FDA Cosmetics Direct. We just track your status.
Serious Adverse Events: You submit via FDA MedWatch (Form 3500A). We generate the package for you to submit.
- Product Safety Vault — Organize labels, safety substantiation docs per SKU
- Adverse Event Log — Track the 15 business-day serious adverse event clock
- Audit Pack Exports — Deterministic ZIPs with manifest (versions + timestamps)
- MoCRA Profile Tracker — Track registration/listing status (no filing)
- SAE Submission Package Export — Generate 3500A-ready packets (no submission)
- Batch Transparency Page (Growth+ plan) — Public pages with disclaimer: "Provided by the brand. Not independently verified."
Ready to simplify your MoCRA recordkeeping?
Join the waitlist today and be among the first Shopify cosmetic brands to experience streamlined compliance documentation.
Join the Waitlist